
To determine the number of neutrons in this isotope, we subtract 26 from 56: 56 − 26 = 30, so there are 30 neutrons in this atom. The 26 is the atomic number (which is the same for all iron atoms), while the 56 is the mass number of the isotope. Which indicates a particular isotope of iron. Atomic numbers are often listed as a subscript on the left side of an element’s symbol. Given the mass number for a nucleus (and knowing the atomic number of that particular atom), you can determine the number of neutrons by subtracting the atomic number from the mass number.Ī simple way of indicating the mass number of a particular isotope is to list it as a superscript on the left side of an element’s symbol. The mass number of an atom is the sum of the numbers of protons and neutrons in the nucleus. When scientists discuss individual isotopes, they need an efficient way to specify the number of neutrons in any particular nucleus. In fact, there are currently over 3,500 isotopes known for all the elements. Most elements exist as mixtures of isotopes. Dalton thought that all atoms of the same element were exactly the same. The discovery of isotopes required a minor change in Dalton’s atomic theory. Figure 2.6 “Isotopes of Hydrogen” compares the three isotopes of hydrogen. An atom is neutral when it contains equal numbers of electrons and protons. An extremely rare hydrogen isotope, tritium, has 1 proton and 2 neutrons in its nucleus. About 1 in 10,000 hydrogen nuclei, however, also has a neutron this particular isotope is called deuterium. Most hydrogen atoms have a nucleus with only a single proton. 
Naturally occurring carbon on Earth, therefore, is actually a mixture of isotopes, albeit a mixture that is 99% carbon with 6 neutrons in each nucleus.Īn important series of isotopes is found with hydrogen atoms. For example, 99% of the carbon atoms on Earth have 6 neutrons and 6 protons in their nuclei about 1% of the carbon atoms have 7 neutrons in their nuclei. Atoms of the same element that have different numbers of neutrons are called isotopes. Astronomy has been associated with the detection of electromagnetic waves or photons from within and beyond the solar system, ranging. However, it was found that atoms of the same element can have different numbers of neutrons. How many neutrons are in atoms of a particular element? At first it was thought that the number of neutrons in a nucleus was also characteristic of an element. The largest atoms have over 100 protons in their nuclei. From this table, you can determine the number of protons in the nucleus of any element. Chapter 21 “Appendix: Periodic Table of the Elements” lists the elements and their atomic numbers. There is no such thing as a hydrogen atom with 2 protons in its nucleus a nucleus with 2 protons would be a helium atom. Helium has the atomic number 2 all helium atoms have 2 protons in their nuclei. 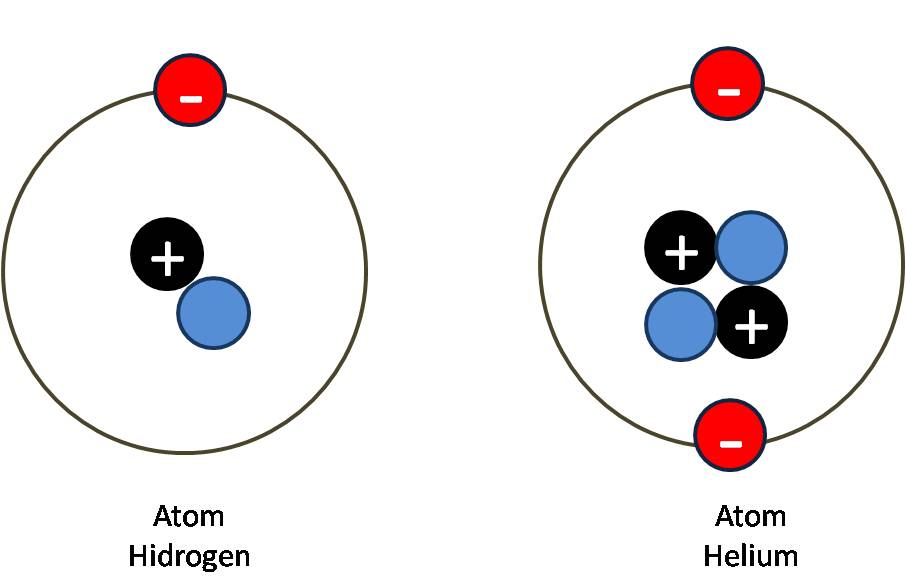
This discovery was so important to our understanding of atoms that the number of protons in the nucleus of an atom is called the atomic number.įor example, hydrogen has the atomic number 1 all hydrogen atoms have 1 proton in their nuclei. Furthermore, different elements have a different number of protons in their nuclei, so the number of protons in the nucleus of an atom is characteristic of a particular element. In other words, all atoms of the same element have the same number of protons. 
The Author(s), under exclusive licence to Springer Nature Limited.In the 1910s, experiments with X rays led to this useful conclusion: the magnitude of the positive charge in the nucleus of every atom of a particular element is the same. These results highlight the emergent capability of neutral-atom qubit arrays for universal, programmable quantum computation, as well as preparation of non-classical states of use for quantum-enhanced sensing. Preparation of entangled Greenberger-Horne-Zeilinger (GHZ) states 5 with up to six qubits, quantum phase estimation for a chemistry problem 6 and the quantum approximate optimization algorithm (QAOA) 7 for the maximum cut (MaxCut) graph problem are demonstrated. Here we demonstrate several quantum algorithms on a programmable gate-model neutral-atom quantum computer in an architecture based on individual addressing of single atoms with tightly focused optical beams scanned across a two-dimensional array of qubits. We know that electrons and protons in an atom have negative and positive charges. Combined with the strong entangling interactions provided by Rydberg states 2-4, all the necessary characteristics for quantum computation are available. A neutral atom is an atom with no charge. Neutral-atom hyperfine qubits provide inherent scalability owing to their identical characteristics, long coherence times and ability to be trapped in dense, multidimensional arrays 1. Gate-model quantum computers promise to solve currently intractable computational problems if they can be operated at scale with long coherence times and high-fidelity logic.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
